EFSA Announces Progress in Pesticide Regulation Updates for Flonicamid and Kresoxim-Methyl
Regulatory progress of flonicamid and kresoxim-methyl issued recently by EFSA.
In June 2024, the European Food Safety Authority (EFSA) launched public consultations regarding two significant pesticide active substances, namely: flonicamid and kresoxim-methyl. These consultations invite the public to provide comments and scientific data, ensuring a comprehensive review process.
As one of the three main Statutory Bodies in Plant Protection Products (PPPs) regulation, EFSA (European Food Safety Authority) works closely with RMS (Rapporteur Member State) on the evaluation of active substances and PPPs, while COM (European Commission) is a decision-maker organization.
To know more about EU Pesticide Regulation, please click here.
Flonicamid and Kresoxim-Methyl
Flonicamid is a pyridine organic compound used as an insecticide in agriculture to control a variety of sucking pests, including aphids, whiteflies, and thrips. Its mode of action involves inhibiting the feeding of these pests, which ultimately leads to their death. Unlike many other insecticides, flonicamid does not kill insects instantly; instead, it disrupts their feeding behavior, reducing damage to crops over time.
Kresoxim-methyl is used in fungicides to control a broad spectrum of fungal diseases in crops such as cereals, fruits, and vegetables. It belongs to the strobilurin class of fungicides and works by inhibiting mitochondrial respiration in fungi, thereby preventing their growth and spread.
The progress of these two substances is significant for the agrochemical industry of the EU.
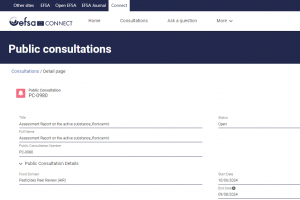
Public Consultation on Flonicamid
EFSA’s Pesticides Peer Review Unit is calling for public comments on the active substance flonicamid. The consultation aims to gather feedback on the Rapporteur Member State’s Assessment Report.
Stakeholders are encouraged to submit their comments by the indicated deadline, ensuring they adhere to the specified format:
- At the beginning of each comment, reference the assessment report by indicating the Volume number, chapter number, page(s), and a brief description (e.g., “Vol. 3, B.8.1, page(s) 41, Route and rate of degradation”).
- Comments should be submitted via the appropriate tool, and not by email.
- All submissions must be copyright-cleared and presented according to EFSA’s guidelines.
To submit your comments, please refer to the detail page of the consultation on EFSA website. Click here to the the documents for consultation.
A parallel consultation on CLH proposal (Proposal for Harmonised Classification and Labelling) for flonicamid is ongoing on the European Chemistry Agency (ECHA) website. Comments related to the CLH report should be directed to ECHA. For more details and to participate, visit the ECHA website.
Public Consultation on Kresoxim-Methyl
ASCENZA AGRO, S.A, BASF SE, INDUSTRIAS AFRASA, S.A., and Lainco S.A. have jointly submitted applications for the renewal of approval of the active substance Kresoxim-Methyl, pursuant to Commission Implementing Regulation (EU) 2020/1740. The dossiers include comprehensive scientific data and studies:
- The lead dossier, submitted by the lead applicant, contains joint information and confidential data.(dossier UUID: b22d67d1-cfcb-4948-9d9e-71348c71fe68)
- Member dossiers include confidential information specific to each applicant.(dossier UUID: 5e4c7212-88a4-4198-a3f3-1331c7789a83 by ASCENZA AGRO S.A; dossier UUID: a7b5afa7-621b-468c-9277-f74af4e7f606 by LAINCO S.A. and INDUSTRIAS AFRASA S.A.)
Stakeholders are invited to review the public dossiers and provide their comments. References to the specific dossier UUID should be included at the beginning of each comment (e.g., “UUID xxx: A new study is available showing toxicity to birds”). Relevant contributions will be reviewed by RMS and EFSA during the peer review process.
To submit your comments, please refer to the detail page of the consultation on EFSA website. Click here to see the attached PDF of the public dossiers (lead and member dossiers).
Submission Guidelines and Deadlines
Comments for both consultations must be submitted by the specified deadlines.
| Flonicamid | Kresoxim-Methyl | |
| Start Date | 10/06/2024 | 13/06/2024 |
| End Date | 09/08/2024 | 12/08/2024 |
Contributions should be finalized, adhere to the provided instructions, and use the relevant digital forms. Please note that comments containing personal accusations, irrelevant or offensive statements, or materials will not be considered. All comments and supporting documents will be published in their original form after the closing date and may be reused by EFSA in different contexts.
Submission Template of the Consultation on Flonicamid
Submission Template of the Consultation on Kresoxim-Methyl
For additional details on participation, visit respective pages on the EFSA website.
Click here to learn more about the Flonicamid consultation.
Click here to learn more about the Kresoxim-Methyl consultation.
Your input is crucial for the thorough evaluation and approval process of these substances.
Contact Us
REACH24H is specialized in helping companies in the pesticide industry comply with global regulations. We offer a range of services, including registration, notification, data support, and compliance services, to keep companies informed about the latest regulatory developments and best practices.
- EU Plant Protection Products Regulation (EU PPP)
- EU Fertilizer Regulation
- EU Biocidal Products Regulation (EU BPR)
- Pesticide Risk Assessment
- GLP Study Monitoring
- QSAR
- Agrochemical Market Research & Regulation Compliance Report
If you need any compliance services, please feel free to contact us.
Email: customer@reach24h.com | Tel: +86 400 809 5809